|
3/25/2023 0 Comments Chlorine atomic numberThe carbon dioxide and limewater react to produce water in addition to the calcium carbonate. What happens when Lime Water reacts to Carbon Dioxide?Ĭarbon dioxide reacts with limewater to form calcium carbonate, which precipitates out of the solution. In this article, we have answered all the questions related to the reaction of lime water and. 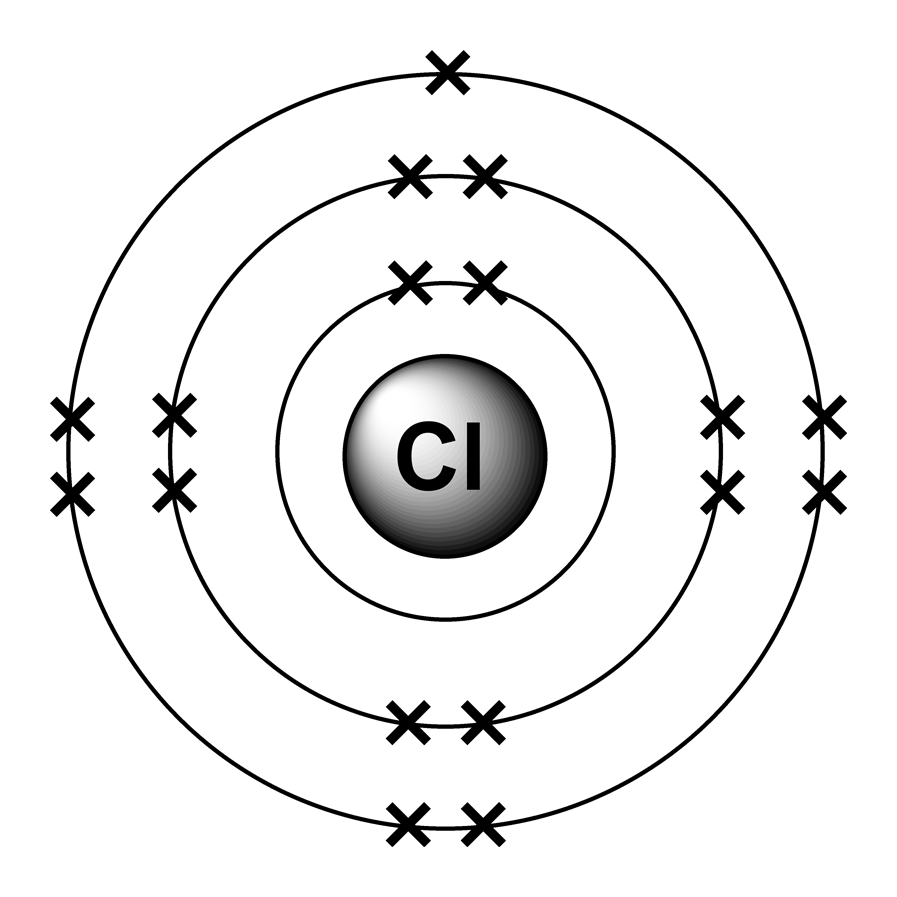
But one of its most noteworthy property is that it is used to absorb carbon dioxide from the air. Limewater is an aqueous solution of slaked lime and you will find it in antacids, medicines and lotions. You may be wondering what is lime water used for. Carbon dioxide is the only gas that turns lime water cloudy. You may often come across a question "What gas turns limewater cloudy?" The answer to this question is well known. Given this information, determine the activation energy for the reverse reaction, Er, and comment on the significance of the value (one sentence only). (iii)The activation energy for the forward reaction, Ef, of step 1 (Equation 3) is 243.4 kJ mol^-1. (ii) With reference to the three-step mechanism (Equations 3–5 ), and assuming that the second step (Equation 4) is rate-limiting, derive the chemical rate equation for this mechanism and then compare it with the experimental rate equation given in Equation 2. (i) Explain why the form of the experimental rate equation indicates that Reaction 1 cannot be an elementary reaction. Under certain experimental conditions, the experimental rate equation was found to be:Ī three-step mechanism has been proposed for Reaction 1: The gas-phase reaction between trichloromethane, CHCl3, and chlorine, Cl2, has time-independent stoichiometry and can be represented as follows:Įquation 1 CHCl3(g) + Cl2(g) = CCl4(g) + HCl(g) I shall stop at this point because this discussion now leads on to writing formulae of ionic compounds! The way I have explained should also help you to write the formulae of the chlorides and oxides, depending on how many electrons the metal is going to lose Group 1 metals lose 1 outer electron, group 2 lose 2 outer electrons. And so reactivity increases with atomic number.3. The outermost electron in potassium is further away from the positive nucleus and so there is less electrostatic force between this outer electron and the positive proton Potassium will therefore be more reactive than Lithium. Which will be more reactive ie which of the two will lose the electron more easily and therefore be more reactive? It has to be potassium. From the info in (1), you will understand that the metal will have to lose electrons for the reaction with chlorine or oxygen Lithium has a electron configuration of 2,1 and potassium 2,8,8,1 ( higher atomic number). Chlorine will react by gaining one electron to form a chloride ion, oxygen react by gaining two electrons to form an oxide ion- reason by gaining electrons they are completing their outer energy level.2. For chlorine it is 2,8,7, and for oxygen it is 2,6. 
To understand why, you need to be able to write the electron configurations of the metal and non metal. Metals react by losing electrons and non metals react by gaining electrons. Points to understand include:1.Reactions between metals and non metals happen because of loss or gain of electrons. But remember where you get those three marks! It's the same across exam boards and comes up practically every year!Ĭhlorine and oxygen are both nonmetals and they react with alkali metals eg lithium and potassium to form chlorides and oxides. Therefore the outer electron is more easily donated to chlorine or oxygen, and so the atom reacts more easily."Or words to that effect. There is also more electron shielding between the nucleus and outer electron, so nuclear attraction decreases down the group. There are more electron shells between the outer shell and nucleus, which causes electron shielding.In an A-level exam you would get 3 marks for saying, "as you go down group 1 the number of electron shells increase, therefore the atomic radius increases. Since each electron shell can only hold so many electrons you increase the number of shells, so the outermost electron is further away from the nucleus with each descending atom, therefore the atomic radius increases (distance between outer electron and nucleus). Since the electrons are negatively charged there is nuclear attraction from the protons.There are always the same number of electrons as protons in neutral atoms, so the number of electrons also increases as you go down group 1. I agree with Daniel, this is a trend seen with the alkali metals and you need to be specific in your answer, or when repeating the question.As you go down Group 1 (alkali metals) the atomic number increases, therefore there are more protons in the nucleus.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
